Applying (R,S) descriptors to biaryls
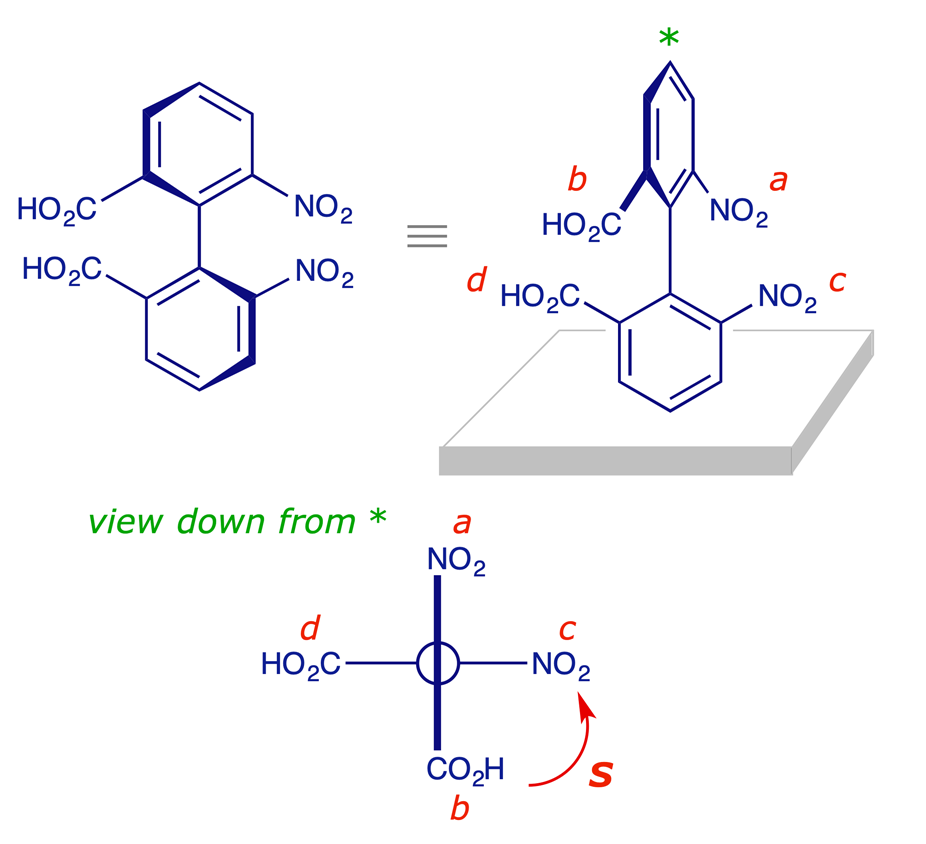
The Cahn-Ingold-Prelog system includes procedures for assigning stereochemical descriptors to molecules with axial chirality, and two examples are provided below. The first is 6,6'-dinitrobiphenyl-2,2'-dicarboxylic acid, and the procedure is applied as follows:
- The molecule is viewed in the conformation with the two aryl rings perpendicular, along the axis of the Ar–Ar bond.
- The four relevant substituents are projected onto a plane at right angles to the axis, and the groups are assigned priorities as though the projection represents a stereogenic atom. It is immaterial whether the plane of the upper ring is seen as vertical (as below) or horizontal.
- The projected atoms or groups may not all be different so the Sequence Rules are augmented with a proximity rule, which is that near groups precede far groups.
(S)-6,6'-dinitrobiphenyl-2,2'-dicarboxylic acid
The proximity rule requires that the vertical (near) groups take precedence over the horizontal (far) groups. Since NO2 precedes CO2H (Sequence Rule), the rear CO2H has the lowest priority d and the remaining groups are prioritised a, b and c as shown, using the Cahn-Ingold-Prelog system. The configuration is (S) because the sequence a to b to c is an anticlockwise turn.
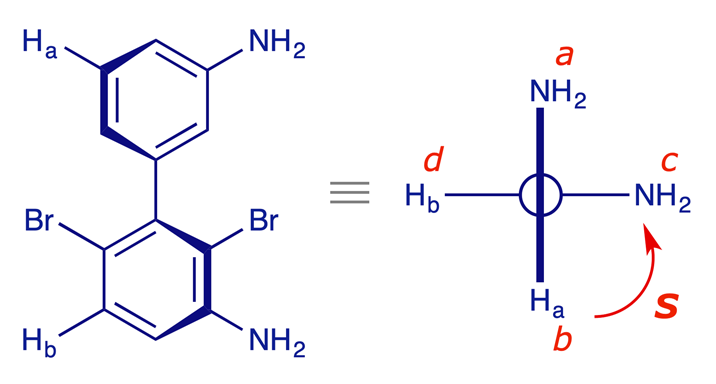
(S)-2,6-dibromobiphenyl-3,3'-diamine
In general for assigning the configuration of a biaryl axis, two pairs of substituents (one pair on each ring, as close together as possible) are chosen, such that the members of each pair can be distinguished by the Sequence Rule. In the example below, the biaryl is shown in the (S)-configuration. Note: The enantiomers of the compound shown are not stable at room temperature – the two bromine atoms alone are not sufficient to prevent rotation about the biaryl bond. But we can assign a configuration to a chiral axis even if it is not stable.